Molarity (M) of a solution is a ratio giving the number of moles of solute per liter of solution.
Molarity (M) = moles of solute (n) / liters of solution (V)
This module is to calculate the molarity of a solution by knowing the moles of the solute directly or indirectly, and the volume of the solution, or calculate either the moles of the solute or the volume of the solution by knowing the molarity and the other quantity.
Question: What is the molarity of a solution prepared by dissolving 50.0 g of KNO3 in enough water to gave 825 mL of solution?
This is one step process, enter the known data and press Calculate to output the unknowns.
1. Select Molarity link from the front page or Molarity tab from the Solution module. The Input and Output screen appears.
2. In the Input area, enter the three known quantities with a proper significant figure. Select the units associated with the input.
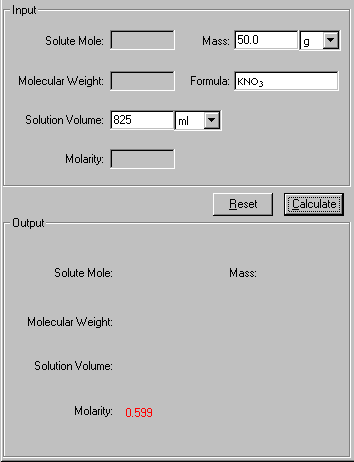
3. Click Calculate to output the answer.
4. The Show Work area on the right shows you step-by-step how your problem has been solved.
To start a new problem, click Reset. All Input fields will be cleared. Follow Step 1-3 again.