Dissociation of Water
The dissociation of a water molecule is the formation of equal amounts of hydronium ion and hydroxide ion:
H2O --> H+ + OH-
And at any time, the product of the hydronium and hydroxide ion molarities in an aqueous solution, at 25°C, must equal to 1.00 x 10-14.
The pH and pOH are the convenient ways to represent molarities of hydronium ion and hydroxide ion.
pH and pOH
The pH and pOH of a solution are defined as:
pH = -log[H+]
pOH = -log[OH-]
and
pH + pOH = 14.00
This module calculates any of three values: pH, pOH, [H+] and [OH-], if any one of them is known.
For example, if the concentration of the hydronium ion in a solution is known as:
[H+] = 1.234x10-5
the [OH-], pH and pOH can be calculated as:
[OH-] = 1.00 x 10-14 / [H+] = 1.00 x 10-14
/ 1.234 x 10-5 = 8.104x10-10
pH = -log[H+] = -log (1.234x10-5) = 4.9087
pOH = 14.00 - pH = 14.00 - 4.9087 = 9.0913
For the above example,[H+] = 1.234x10-5, calculate pH.
This is one step process, enter the known data and press Calculate to output the unknowns.
1. Select Interconversion link from the front page or Interconversion tab from the Acid, Baseand Salt module. The Input and Output screen appears.
2. In the Input area, enter the known quantities with a proper significant figure.
3. Click Calculate to output the answer.
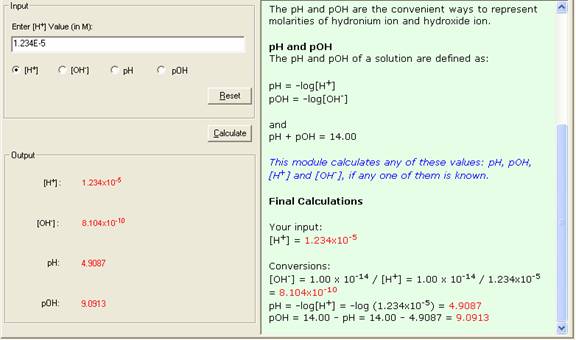
4. The Show Work area on the right shows you step-by-step how your problem has been solved.
To start a new problem, click Reset. All Input fields will be cleared. Follow Step 1-3 again.