Titrations
The amount of acid or base in a solution can be determined by titration.The process of titration involves a solution of known concentration (titrant) delivered from a buret into the solution of unknown concentration until the stoichiometric point is achieved. The key point in acid-base titration is to calculate the pH of the solution when different amount of the titrant are added. A pH curve is a plot of the pH changes in the solution as a function of the amount of titrant added.
There are three types of Acid-Base titration:
The millimole (abbreviated mmol) is very common unit used in titrations:
mmol = Molarity x mL solution = 10-3 mol
This module is for one of the three possible cases of titration. By choosing a titrant of known concentration and an unknown solution, the key numbers (initial pH, pH at the equilibrium point and the volume needed to get the equilibrium) and pH curves can be calculated.
Strong Acid-Strong Base Titrations
In the titration of 80.0mL of 0.300M HNO3 with 0.200M NaOH,
Initial moles of H+ = 24.0mmol
Added OH- moles = 120.0mL x 0.200M = 24.0mmol
At this point, enough OH- has been added to react completely with the H+ from the HNO3, so the solution is neutral (pH = 7.00)
This is one step process, enter the known data and press Calculate to output the unknowns.
1. Select Strong Acid/Strong Base link from the front page or Strong Acid/Strong Base tab from the Acid, Baseand Salt module. The Input and Output screen appears.
2. In the Input area, enter the known quantities with a proper significant figure with the above example.
3. Click Calculate to output the answer.
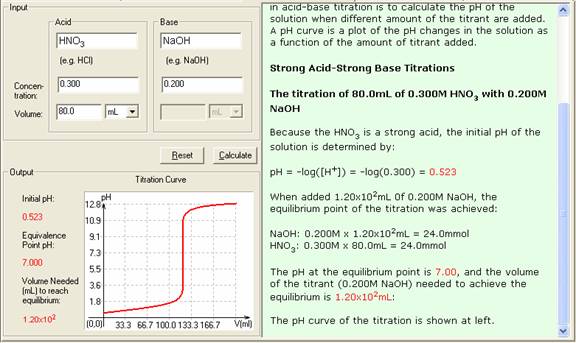
4. The Show Work area on the right shows you step-by-step how your problem has been solved.
To start a new problem, click Reset. All Input fields will be cleared. Follow Step 1-3 again.